Dec 2022
From form to function ? pre- and post-synthetic functionalization as the method of synthesis of chiral, shape-persistent macrocycles
Chemistry Graduate Seminar
Speaker: Professor Marcin Kwit, Laboratory of Organic Stereochemistry, AMU
Abstract
Since the early discoveries by Ru?i?ka,[1] and the award of the Nobel Prize to Cram, Lehn and Pedersen in the field of chemistry in 1987, there has been observed a continuous increase in the interest in artificial macrocyclic compounds, which have become one of the most important compounds in chemistry, material chemistry, crystal engineering and biochemistry. Macrocycles constitute fragments of some molecular devices capable of performing mechanical work under an influence of external stimuli. The most relevant examples of macrocycles, playing a crucial role in chemistry and/or of high biological activity, include, among others, cyclodextrins, porphyrins, antibiotics, macrolides, anticancer and other therapeutic agents (Figure 1a).[2]
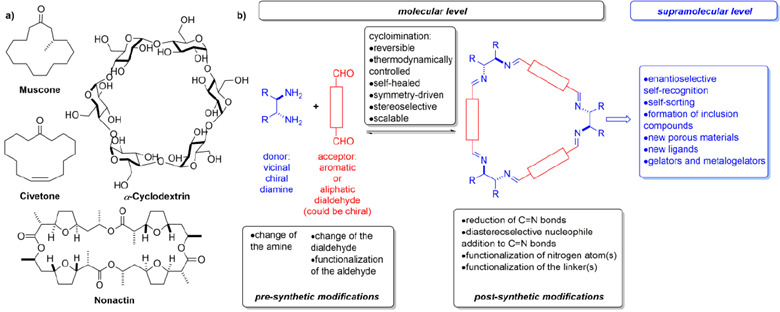
Figure 1. a) Examples of naturally-occurring macrocycles b) Key points of the macrocyclization reaction with the specified main features of the building blocks and products.
Despite current progress, an easy access to large molecules of defined structure, predictable and modifiable properties and applicable as functional molecules or components of functional materials is still a challenging task.[3]
The use of thermodynamically controlled (dynamic) reversible reactions gives a chance to obtain compounds with high yields, if the energy gain associated with the formation of cyclic products overwhelms that of acyclic oligomers. Among the reactions usually considered compatible with Dynamic Covalent Chemistry (DCvC) rules, the imination reaction is of a special position, as in the modern manner, the reaction might provide complex products from structurally predisposed building blocks (Figure 1b).[4]
Considering the cyclization as the turning point in synthesis, the building blocks, as well as the products, might be functionalized either at the pre- or post-synthetic stage. The modular structure of polyimine macrocycles allow, in principle, for almost unlimited modifications of their properties, i.e., by expansion of the basic structure in the direction parallel to the main symmetry axis of the molecule or by increasing the macrocycle cavity.[5] However, except for single examples, the potential for such derivatization has not yet been exploited. Due to the high sterical congestion, the “overcrowded” macrocycles having bulky substituent(s) in very close proximity to the newly formed imine bond(s) remain unknown.
In this lecture, I will prove that intentionally designed selected group of chiral macrocyclic compounds of periodical, symmetrical and modular structure, is still challenging synthetic target and the compounds, when obtained, may perform diverse function. The mechanism of an action of these compounds is based on some fundamental processes, namely molecular recognition, sorting, aggregation and assembly. The synergistic (cooperative) effects, exerted by molecular fragments present within the same molecule, can be particularly useful in stereoselective synthesis. The use of macrocycles as molecular synthons (tectons) in molecular tectonics allows for modeling capability of inclusion and/or porosity of solids.
Additionally, the initially obtained macrocyclic structures may constitute a starting point for the synthesis of molecules impossible to obtain by other reactions.
[1] L. Ru?i?ka, M. Stoll, H. Schinz, Helv. Chim. Acta 1926, 9, 249.
[2] see for example: a) Z. Liu, S. K. M. Nalluri, J. F. Stoddart, Chem. Soc. Rev. 2017, 46, 2459; b) T. Ogoshi, T. Yamagishi, Y. Nakamoto, Chem. Rev. 2016, 116, 7937; c) C. Gropp, B. L. Quigley, F. Diederich, J. Am. Chem. Soc. 2018, 140, 2705.
[3] a) V. Martí-Centelles, M. D. Pandey, M. I. Burguete, S. V. Luis, Chem. Rev. 2015, 115, 8736; b) L. A. Wessjohann, D. G. Rivera, O. E. Vercillo, Chem. Rev. 2009, 109, 796.
[4] a) N. E. Borisova, M. D. Reshetova, Y. A. Ustynyuk, Chem. Rev. 2007, 107, 46; b) Y. Jin, C. Yu, R. J. Denman, W. Zhang, Chem. Soc. Rev. 2013, 42, 6634; c) S. Srimurugan, P. Suresh, B. Babu, H. N. Pati, Mini-Rev. Org. Chem. 2008, 5, 228.
[5] M. Kwit, J. Grajewski, P. Skowronek, M. Zgorzelak, J. Gawronski, Chem. Rec. 2019, 19, 213.
Bio
Marcin Kwit was graduated with honors from the Department of Chemistry of Adam Mickiewicz University in 1998. After receiving his PhD degree at the Adam Mickiewicz University in 2003 under supervision of Professor Jacek Gawro?ski, he joined the research group of Prof. Ben L. Feringa at the University of Groningen, where he worked on molecular machines and switches. After having moved back to Poland, Marcin Kwit has set-up his independent research group. In 2012 the Council of the Department of Chemistry of Adam Mickiewicz University awarded him the degree of postdoctoral (habilitation) PhD in chemical science in the discipline of chemistry. The postdoctoral thesis "Determination of the absolute configuration of conformationally labile compounds" was the basis for his postdoctoral application.
Marcin Kwit was appointed to the position of Head of the Laboratory of Organic Stereochemistry in 2013. Since 2020 he is a Full Professor of Organic Chemistry at the Adam Mickiewicz University in Pozna?. In the meantime, he was appointed as Assistant Professor in the Institute of Organic Chemistry Polish Academy of Sciences, Warsaw.
His research interests include stereochemistry of conformationally labile compounds, circular dichroism spectroscopy, the chemistry of chiral macrocycles and covalent organic cages, the use of molecular propellers as chromophoric probes and structural elements in supramolecular systems and applications of macrocyclic compounds and triarylmethane derivatives as ligands or organocatalysts in asymmetric synthesis.
Marcin Kwit has received several awards including the Scholarship of the Minister of Education of Poland and the Prime Minister's Award.
Since 2021 he is a visiting professorial fellow of the Northwest Normal University, Lanzhou, China.